Description
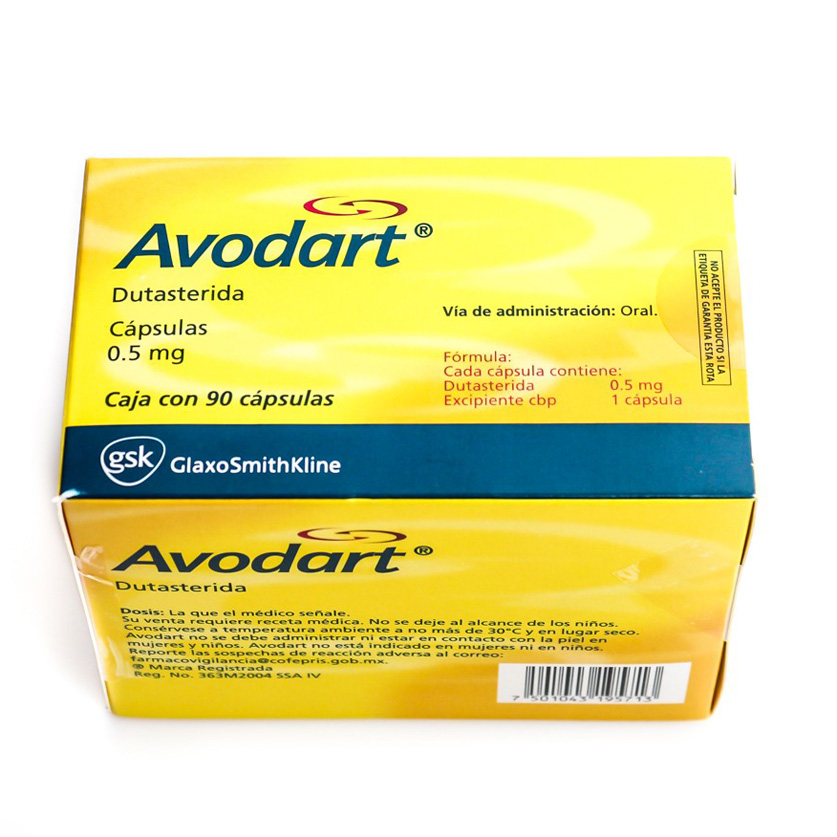
Dutasteride (INN, USAN, BAN, JAN) (trade name Avodart), manufactured by GlaxoSmithKline, is a triple inhibitor of 5? -reductase which inhibits the conversion of testosterone to dihydrotestosterone (DHT). It is used to treat benign prostatic hyperplasia. It increases the risk of erectile dysfunction and decreases sexual desire. Medical uses [edit]
Avodart (dutasteride) 500 μg soft capsules
Dutasteride is useful for the treatment of benign prostatic hyperplasia (BPH); colloquially known as "enlarged prostate".
In those who are regularly screened, 5-reductase inhibitors such as finasteride and dutasteride reduce the overall risk of being diagnosed with prostate cancer; however, there is insufficient data to determine whether they have an effect on the risk of death and may increase the likelihood of more severe cases.
Side effects
Sexual effects
This class of drugs increases the rates of erectile dysfunction (between 5% and 9% develop problems after starting their use). This is related to the lower quality of life and can cause stress in relationships. There is also an association with lowered sexual desire. It has been reported that these negative sexual side effects can persist even after discontinuation of the drug.
Prostate Cancer
The FDA added a warning to dutasteride about an increased risk of high-grade prostate cancer. While the potential for positive, negative, or neutral changes in the potential risk of developing prostate cancer with dutasteride has not been established, evidence has suggested that it may temporarily reduce the growth and prevalence of benign prostate tumors, but it may also mask the early diagnosis of prostate cancer. The main area of concern is for patients who may develop prostate cancer while taking dutasteride for benign prostatic hyperplasia, which in turn could delay early diagnosis and treatment of prostate cancer, potentially increasing the risk of these patients develop high-grade prostate cancer.
Contraindications
Children and women who are or could become pregnant and people with known significant hypersensitivity (eg, severe skin reactions, angioedema) to dutasteride or finasteride should not take dutasteride. Exposure to dutasteride and other 5-reductase inhibitors during pregnancy can cause birth defects. Since these drugs are easily absorbed through the skin, women who are or could become pregnant should not handle them and if they come into contact with leaking capsules, the contact area must be washed immediately with soap and water. People taking dutasteride should not donate blood, and due to its long half-life, they should not donate blood for at least 6 months after stopping treatment.
Mechanism of action
Dutasteride belongs to a class of drugs called 5-reductase inhibitors, which block the action of 5-reductase enzymes that convert testosterone into dihydrotestosterone (DHT).
Dutasteride versus finasteride
Finasteride is also approved for the treatment of benign prostatic hyperplasia or BPH. Drugs belong to the same class of drugs.
Dutasteride inhibits all three isoforms of 5-reductase, type I, II and III, while finasteride inhibits only types II and III and has a much shorter half-life.

Reviews
There are no reviews yet.